Over the past several decades there has been a growing awareness of mercury in commercially available fish. In fact, mercury levels have climbed 30 percent in the last 20 years in the Northern Pacific alone. Pregnant women, for example, are admonished to only consume a certain amount of fish. However, little thought is given to how the mercury ended up in the water in the first place.

The path of mercury from its primary sources in power plants, into the water, and then into our bodies through both drinking water and eating fish, is a curious one. It is also one that we must understand if we are to come up with sensible solutions for the very real problem of mercury contamination in our oceans, rivers, and lakes.
It is not necessary to be a climate doomsayer or a green fanatic to understand the importance of keeping toxic chemicals out of the water. In fact, an outsized emphasis on global temperature changes can divert attention away from arguably more pressing issues like mercury in the water.
Indeed, there are very sensible solutions to this problem that do not require massive reductions in the Western standard of living or unreliable and arcane technologies not ready for prime time.
Just How Bad Is Mercury in the Environment?

Because we are so inundated with environmental doomsaying, it can be hard to know what is worth worrying about and what is simply noise. Mercury in the air, land, and water is certainly a very real concern, particularly with regard to the contamination of seafood, which makes up a significant protein source for billions of people around the world.
But if this weren’t disturbing enough, there is also mercury in the water that we drink. Fortunately, it is possible to remove the mercury from your water supply using reverse osmosis filters, distillation, GAC filters, and sub-micron filters.
The effects of mercury consumption are not abstract nor are they anything to trifle with. For example, you probably know that women are supposed to avoid eating too much fish during pregnancy because of mercury contamination.
What you might not know, however, is that it is estimated that 6% to 8% of the 16-to-49-year-old women in America have critical levels of mercury in their bloodstream. For certain segments of the population, this percentage jumps to 15 percent.

We have no method of removing mercury from the fish, however. All we can do is avoid the more dangerous fish (or eat them sparingly) and work to ensure that less mercury is emitted into the environment in the first place. The latter is an arduous task, but there is much reason to be hopeful that the momentum is going in the right direction.
Finally, fish and drinking water are not the only ways that we come into contact with dangerous levels of mercury. Many thermometers still contain mercury and are a source of poisoning for many who continue to use broken thermometers.

Mexican novelty jewelry is known to contain mercury, as are a number of other consumer products such as barometers and switches, but also in a growing number of cosmetics. Gold mining is another source of exposure and the fillings in your teeth might also be slowly giving you mercury poisoning — it can no longer be dismissed as an urban legend.
The effects of mercury poisoning can easily be debilitating, causing a loss of sight and hearing, or even fatal. Palliative treatments can be made, but mercury poisoning is a very serious illness that should be avoided at all costs.

Perhaps most troubling, exposure to mercury in infancy or in utero can lead to developmental problems that follow one throughout life. This is why it is absolutely essential for new and expecting mothers to err on the side of caution when it comes to consuming fish, particularly those known to be vectors for mercury poisoning.
This might all sound a little far-fetched, but the most common cause of mercury poisoning is the overconsumption of contaminated fish. Hundreds of thousands of pregnant women are impacted by this every year, to say nothing of how many others are suffering from more generalized symptoms of mercury poisoning without knowing it.

If you think you have symptoms of mercury poisoning, a quick audit of your diet might be one of the fastest ways to know if this is a reasonable cause of your symptoms. If so, consider seeing a doctor and submitting a urine test to determine the amount of mercury in your bloodstream.
Where Does the Mercury Come From?
Mercury comes from three primary sources. The first is anthropogenic, or mercury that is emitted into the environment as a result of human activity. The primary vector for mercury in the environment through anthropogenic sources is coal-fired power plants, with this accounting for 50 percent of mercury emissions in the United States.

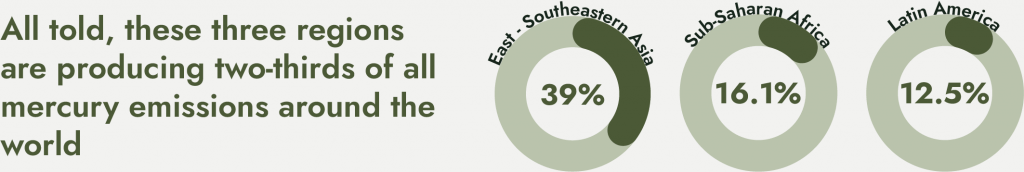
However, the bulk of mercury is emitted into the environment by economically poor countries. East and Southeastern Asia account for 39 percent of all mercury emissions around the world, with Sub-Saharan Africa accounting for 16.1 percent and Latin America accounting for 12.5 percent. All told, these three regions are producing two-thirds of all mercury emissions around the world.
The mercury in the coal is natural, not man-made. When the coal is taken out of the ground, a lot of other materials are taken out with it, both in the coal and as a result of extracting the coal. Mercury is one of those “other materials” that come out with coal.
The path from coal-fired power plants is relatively straightforward: The mercury is emitted into the air, where it enters the water cycle and is rained into the oceans, rivers and streams. However, there is far more mercury than we can account for in the fish. This is how we know that fish are getting their mercury from sources other than coal-fired power plants. This brings us to the second form of mercury emissions.
The second source is natural sources of mercury, which account for about 10 percent of all the mercury in the environment. Volcanos, forest fires, and ore are all sources of mercury in the environment that are not man-made.

In fact, because there is naturally occurring mercury in the environment, your body is capable of processing very small amounts without issue. The problem is when the mercury concentration becomes too high or concentrated.
The biggest source of mercury, however, is one that you likely hadn’t considered before now: reemissions.
This is when mercury, which has already been introduced into the environment by human activity, is re-released by other activity, be it human or otherwise. This accounts for the remaining 60 percent of mercury emissions.

Remissions are worth taking a second to consider because they’re a sort of compounded interest with regard to mercury emissions. The mercury is emitted from its original source, locked somewhere in the environment, re-released, and then locked somewhere else again. So the problem of reemissions is one that is far greater than the problem of original emission — and it accounts for nearly two-thirds of all emissions.
Fortunately, mercury reemissions represent one of the easiest ways to make a dent in mercury in the air and water. Various emerging technologies make it easier to control the amount of mercury released in the first place, as well as control the amount of mercury that is reemitted once it is absorbed into things like limestone and gypsum.
Mercury in the fish also comes from another unlikely source: aquaculture and farmed fish. Farmed fish are often fed food that has been treated with mercury. This makes them contaminated but also means that there is effectively mercury being dumped into the water, to say nothing of mercury emissions from the fish food plants and the emissions from the power plants needed to fuel them.

It is somewhat ironic that the fish most detached from nature are sometimes the most mercury-contaminated fish of all.
There is an additional problem with regard to mercury and our oceans: Once the water enters the ocean, it is chemically transformed into a more dangerous form of mercury known as methylmercury. This means that by the very fact of entering the water, the mercury has become far more deadly to our bodies than it was simply floating around in the air.
In fact, there’s an area of the ocean where the mercury collects in greater concentration than it does in other areas of the ocean: Between 100 and 400 meters thick lying between 100 and 1000 meters below the surface. No one is entirely sure why, but experts suspect that it has something to do with the bacteria that consume and emit mercury in the ocean.

Biomagnification: The Vector for Mercury Into Your Body
Next, we have to understand the concept of biomagnification. Because the water is full of lots of things, but they don’t all end up in our food. What’s more, when they do, they don’t necessarily come in amounts that are toxic. And, indeed, the actual mercury in the water isn’t terribly toxic to you or me or the fish that swim in it.
The problem comes when we remember how the food chain works. Plankton have a little bit of mercury in them, shrimp eat a bunch of plankton, now the shrimp have more mercury than just one little plankton. Shrimp is one of the biggest offenders for mercury content, but what about things that eat shrimp? Or the fish that eat those fish? And so on.
At every level of the food chain, the amount of mercury in the fish is magnified, sometimes many fold. By the time you’re getting up to fish the size of swordfish, you’re talking about probably thousands of different organisms in the food chain that were contaminated with mercury.

This is why larger predator fish like swordfish and tuna are some of the most mercury-contaminated fish on the shelves of our supermarkets today.
The bigger the fish, the more its diet includes smaller organisms that also are carnivorous, the more mercury it is likely to have accumulated from the sum total of the food chain. Knowing this can act as a rough guide if you are unsure about the general mercury content of a specific kind of fish, but you should always do your own research.
Methods of Controlling Mercury
We have several methods of controlling mercury escaping into the environment in the first place that doesn’t mean shuttering every coal-fired power plant in the nation.
Bromine, for example, is a chemical that can be introduced into coal-fired power plants that will reduce the amount of mercury released into the atmosphere significantly.
There are other methods of preventing mercury from reaching the streams and oceans in the first place. First, there are emerging technologies that mean that coal-fired power plants are far more efficient, meaning that they produce more energy while emitting less of everything, including mercury. Natural gas produces less mercury than coal and other alternative fuel sources have as much mercury as coal, but the mercury is easier to control.
Consumer choices can be made, but it can be difficult to know what the right move is. For example, on the one hand, low-energy light bulbs generally contain mercury, but significantly reduce the amount of mercury released into the atmosphere by coal-fired power plants.
At the very least, any products in your home such as batteries, thermometers and thermostats which might contain mercury need to be disposed of properly and not just thrown out with the rest of the trash.

Once the mercury goes into the air, there are a variety of ways to control where it goes and how much of it reaches the atmosphere and the oceans. How effective these techniques are is generally reliant upon the type of coal that is being burned. So-called “end of pipe” techniques seek to reduce the emissions leaving the power plant, but also to contain it.
The main shortcoming of these techniques is that it kicks the can down the road — the mercury that is captured still needs to be disposed of. However, the mercury can now be disposed of in a thoughtful and purposeful way, rather than just being belched into the air.
This leads to the need for effective waste management techniques to dispose of the mercury technicians are able to recapture. Mercury can be treated in small amounts so that it can be disposed of in regular landfills with minimal risk.
The Good News About Mercury Emissions
We actually have quite a bit of good news with regard to mercury emissions in the United States: In 2012, the government set a target of reducing mercury emissions by 90 percent.

These efforts have largely been successful, both inasmuch as they have led to less mercury in the environment, but also because the emissions reductions have taken place over a long enough period of time that we can evaluate their health effects.
While the statistics we have might not sound impressive, it’s important to note that these were the effects of less than ten years of a rigorous and coordinated attempt to dramatically reduce mercury emissions: Children born with exposure to mercury declined by half, the average mercury blood level in women declined by over a third, and the measurable mercury levels in lakes, rivers, streams and the land have declined appreciably — all this from an 85 percent reduction in environmental mercury from coal-fired power plants alone.



Bromine does this not just through a direct reduction of the mercury content in emissions. It also converts the mercury from its usual form into one that is much easier to be captured by environmental scientists.
Coal-fired power plants are the main place we should look to reduce mercury emissions, simply because they are the origin of most of the mercury emitted in the United States. It is not necessary to shutter these plants, simply to leverage emerging technologies to contain and reduce the emissions of mercury. Until nuclear power becomes widespread, we are reliant as a society on these power plants.
On the other side of the equation, rising planetary temperatures are making it harder to control re-emissions. But while there is overlap between global warming and mercury emissions, the latter generally takes a backseat in the public consciousness. Why this is is anyone’s guess, but carbon emissions and the debate over climate change continue to hog the spotlight while we have absolutely no debate at all as to whether or not mercury in the environment is harmful in the here and now, not in 20 years.

While we might not be able to agree on a solution for climate change, the solution for mercury contamination is clear. What’s more, the technology is not waiting to be developed, it’s already on the market waiting to be deployed. It’s a massive money-making opportunity and thus, is entirely feasible and likely to be successful because it appeals to the basic human emotion of greed.
What To Buy At The Supermarket
When you go to the supermarket to pick up fish, it’s important to limit your consumption of the most contaminated species of commercially available fish. These are, again, larger predatory fish like shark, ray, swordfish, and tuna — though, despite what you might have heard, canned tuna is one of the safer options on the market. Other less toxic options for your dinner table include virtually all forms of shellfish, salmon, and the aforementioned canned tuna.
There are a number of readily available guides for shopping at the supermarket for fish. The main thing you need to remember is that the more fish that a fish eats, the more mercury it is likely to have. Bluefish, marlin, shark, swordfish, certain types of tuna, mackerel and other forms of large carnivorous fish ought to be avoided entirely. Unfortunately, unlike with other forms of food, one cannot simply make healthier choices by switching brands or similar.
One way that you can make sure that otherwise, iffy fish are slightly safer is by buying them fresh and caught here in the United States. Because we have more stringent environmental laws, this will give you some movement toward less mercury. However, we share an ocean with the entire world and there is only so much that can be done to keep American waters safe when the rest of the world isn’t doing its part.

As far as keeping safe eating what you catch, the best thing you can do is make a call to your local fish and game office and inquire about the safety of the fish in the area where you plan on angling. They’ll be able to provide you with the most accurate, specific and timely information when it comes to whether or not it’s safe to eat what you have pulled out of the water.
While the situation can be daunting, the good news is that the moment is all toward less and less mercury in the water, meaning less and less mercury in the fish.
Make the right consumer choices, be a responsible citizen when disposing of mercury and support political policies that reduce the amount of carbon being emitted by coal-fired power plants and other sources.
While the perfect solution might not be coming down the pike tomorrow, lots of progress is being made. We can confidently say that the problem of mercury contamination will, in the near future, be a thing of the past.




It is so refreshing to read an article about environmental contamination that is not characterized by fear mongering and doomsday scenarios. I learned a great deal from this article and intend to act on the information. I have forwarded it to friends whom I know will appreciate the level-headed tone in which it is written as well. Thank you so much.
Gunnar, we really appreciate the kind words and are very thankful for readers like you who understand the objective of this type of content. We’re all so sick of the fear mongering and ridiculous extremes many “information” outlets will go to. Sometimes it pays to read between the lines! Thanks so much for reading and sharing!
Many articles on mercury do not mention that all small fish are safer and highest in omega3 such as sardines and small mackerel. Also natural detox herbs and algae that can bind mercury and clear it from body. I am concerned that farm fishing is legal and dont provide mercury levels on packaging.